First report of biovar 6 in birds immunized against <em>Gallibacterium anatis</em> in poultry farms located in Sonora, Mexico.
Main Article Content
Abstract
Veterinaria México OA
ISSN: 2448-6760
Cite this as:
- Osuna Chávez RF, Molina Barrios RM, Hernández Chávez JF, Robles Mascareño J, Icedo Escalante JGA, Acuña Yanes M. First report of biovar 6 in birds immunized against Gallibacterium anatis in poultry farms located in Sonora, Mexico. Veterinaria México OA. 2017;4(3). doi: 10.21753/vmoa.4.3.389.
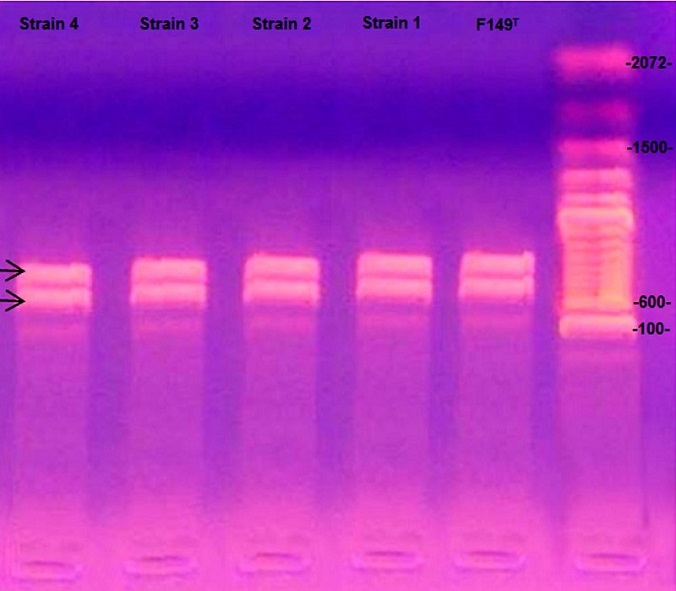
Identification of the predominant circulating biovars is important for controlling Gallibacterium anatis infections in poultry because efficient protection is based on the use of biovar-specific immunogens. Twenty-three isolates of Gallibacterium anatis were recovered from commercial laying hens with respiratory and reproductive clinical signs. Bacteria were identified as G. anatis based on morphology, biochemistry, and PCR testing. Biochemical analyses identified the isolates as biovar 6 which had not previously been reported in Sonora, México. Therefore, the recent finding of biovar type 6 could explain clinical presentations of this disease in immunized commercial laying flocks in Sonora.
Article Details
References
Barrientos Rangel B. Bacteriologia en el complejo respiratorio de las aves | Foros [Technical reports]. SENASICA-SAGARPA; 2011 [updated 24-04-2011].
Castellanos L, Vázquez M, Carrillo A, Campogarrido R. Protección conferida por una vacuna comercial contra Gallibacterium anatis en aves de postura comercial. Boehringer Ingelheim Vetmédica. 2006;5(11):1-3.
Christensen H, Bisgaard M, Bojesen AM, Mutters R, Olsen JE. Genetic relationships among avian isolates classified as Pasteurella haemolytica, ‘Actinobacillus salpingitidis’ or Pasteurella anatis with proposal of Gallibacterium anatis gen. nov., comb. nov. and description of additional genomospecies within Gallibacterium gen. nov. Int J Sys Evol Microbiol. 2003;53:275-87. doi: 10.1099/ijs.0.02330-0.
Bojesen AM, Torpdahl M, Christensen H, Olsen JE, Bisgaard M. Genetic diversity of Gallibacterium anatis isolates from different chicken flocks. J Clin Microbiol. 2003;41:2737-40. doi: 10.1128/JCM.41.6.2737-2740.2003.
Mushin R, Weisman Y, Singer N. Pasteurella haemolytica found in the respiratory tract of fowl. Avian Dis. 1980;24:162-8. doi: 10.2307/1589775.
Mirle C, Schoengarth M, Meinhart H, Olm U. Studies into incidence of Pasteurella haemolytica infections and their relevance to hens, with particular reference to diseases of the egg-laying apparatus. Monatshefte fuer Veterinaermedizin (Germany, FR). 1991.
Jaworski MD, Hunter DL, Ward AC. Biovariants of isolates of Pasteurella from domestic and wild ruminants. J Vet Diagn Invest. 1998;10:49-55. doi:10.1177/104063879801000109.
Bisgaard M, Korczak BM, Busse HJ, Kuhnert P, Bojesen AM, Christensen H. Classification of the taxon 2 and taxon 3 complex of Bisgaard within Gallibacterium and description of Gallibacterium melopsittaci sp. nov., Gallibacterium trehalosifermentans sp. nov. and Gallibacterium salpingitidis sp. nov. Int J Syst Evol Microbiol. 2009;59(4):735-44. doi: 10.1099/ijs.0.005694-0.
Bojesen AM, Nielsen SS, Bisgaard M. Prevalence and transmission of haemolytic Gallibacterium species in chicken production systems with different biosecurity levels. Avian Pathol. 2003;32:503-10. doi: 10.1080/0307945031000154107.
Lowenstine LJ. Necropsy procedures. In: Harrison JG, Harrison LR, editors. Clinical avian medicine and surgery. London (UK): Elsevier - Health Sciences Division; 1986. p. 298-309.
Woodford MH, Keet DF, Bengis RG. Post-mortem procedures for wildlife veterinarians and field biologists: IUCN Lybrary System, Office International des Épizooties; 2000 2000. 55 p.
Carter G, Cole JJ. Diagnostic procedure in veterinary bacteriology and mycology. 5th ed: Academic Press; 1990 28 Jun 1990. 620 p.
Diallo IS, Frost AJ. Characteristics of a haemolytic extract from avian Pasteurella multocida. Vet Microbiol. 2000;72:37-45. doi: 10.1016/S0378-1135(99)00185-6.
Castillo G, Koga Y, Alvarado A, Tinoco R, Fernández D. Aislamiento e identificación bioquímica de cepas de Pasteurella multocida y Gallibacterium anatis en aves de producción con signos respiratorios. Rev Investig Vet Peru. 2014;25(4):516-22. doi: 10.15381/rivep.v25i4.10812.
Myrvik QN, Weiser RS. Bacteriología y micología médicas. 2nd ed. México: Interamericana-McGraw-Hill; 1991 1991. 713 p.
Rimler RB, Sandhu TS, Glisson JR. Pasteurellosis, infectious serositis, and Pseudotuberculosis. In: Swayne DE, editor. A Laboratory Manual for the Isolation and Identification of Avian Pathogens. Philadelphia (US): American Association of Avian Pathologists; 1998.
Benson DA, Karsch-Mizrachi I, Lipman DJ, Ostell J, Wheeler DL. GenBank: update. Nucleic Acids Res. 2004;32:D23-D6. doi: 10.1093/nar/gkh045.
Bojesen AM, Robles F, Vazquez MA, Christensen H, Gonzalez C, Soriano-Vargas E, et al. Specific identification of by a PCR using primers targeting the 16S rRNA and 23S rRNA genes. Vet Microbiol. 2007;123(1-3):262-8. doi: 10.1016/j.vetmic.2007.02.013.
Lane DJ. 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M, editors. Nucleic acid techniques in bacterial systematics. Michigan (US): Wiley; 1991.
Neubauer C, De Souza-Pilz M, Bojesen A, Bisgaard M, Hess M. Tissue distribution of haemolytic Gallibacterium anatis isolates in laying birds with reproductive disorders. Avian Pathol. 2009;38:1-7. doi: 10.1080/03079450802577848.
Tegtmeier C, Angen Ø, Ahrens P. Comparison of bacterial cultivation, PCR, in situ hybridization and immunohistochemistry as tools for diagnosis of Haemophilus somnus pneumonia in cattle. Vet Microbiol. 2000;76:385-94. doi: 10.1016/S0378-1135(00)00259-5.
License

Veterinaria México OA by Facultad de Medicina Veterinaria y Zootecnia - Universidad Nacional Autónoma de México is licensed under a Creative Commons Attribution 4.0 International Licence.
Based on a work at http://www.revistas.unam.mx
- All articles in Veterinaria México OA re published under the Creative Commons Attribution 4.0 Unported (CC-BY 4.0). With this license, authors retain copyright but allow any user to share, copy, distribute, transmit, adapt and make commercial use of the work, without needing to provide additional permission as long as appropriate attribution is made to the original author or source.
- By using this license, all Veterinaria México OAarticles meet or exceed all funder and institutional requirements for being considered Open Access.
- Authors cannot use copyrighted material within their article unless that material has also been made available under a similarly liberal license.



