Ultrastructure of bovine preantral follicles: effect of the slaughterhouse aseptic process on follicular integrity
Main Article Content
Abstract
Veterinaria México OA
ISSN: 2448-6760
Cite this as:
- Cisneros Prado JL, Montiel Palacios F, Canseco Sedano R, Merchant Larios H. Ultrastructure of bovine preantral follicles: effect of the slaughterhouse aseptic process on follicular integrity. Veterinaria México OA. 2017;4(2). doi: 10.21753/vmoa.4.2.424.
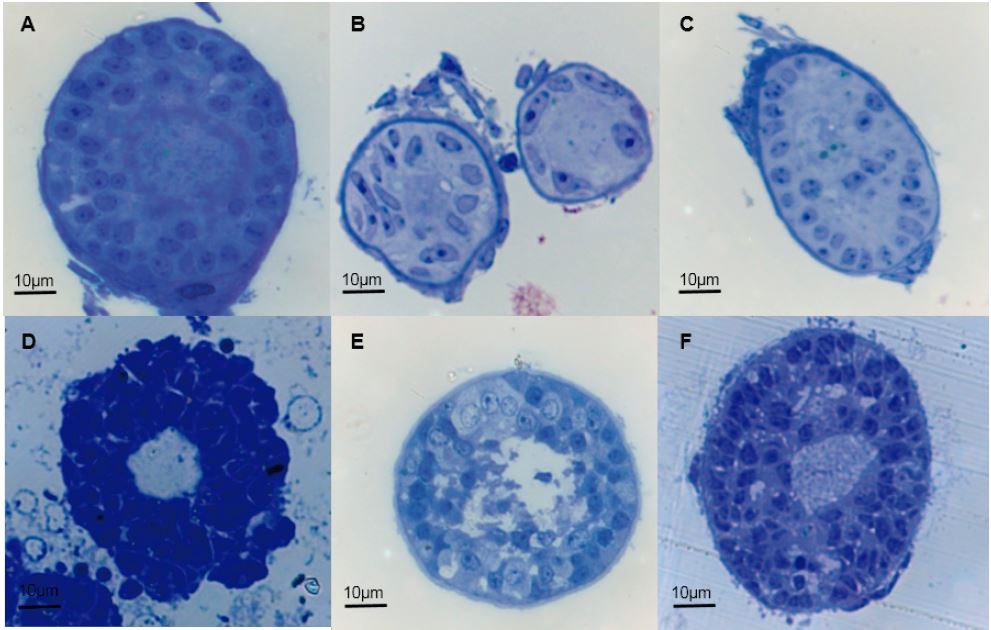
Ovaries from slaughtered cows are the most abundant source of preantral immature follicles for in vitro studies including maturation and in vitro fertilization. Integrity of immature follicles may be compromised by inadequate management during the collection and transport of the gonads, limiting the success of a correct evaluation. This study assessed the effect of aseptic and transport of bovine ovaries on the structural and ultrastructural integrity of immature follicles from the ovarian cortex. Ovaries from 20 different crossbred cows (Bos taurus × B. indicus) were randomly assigned to one of two aseptic and transport treatments: (1) the ovaries were washed once with 70 % ethanol for 10 s, rinsed twice with normal saline solution (OH + NSS), and then transported in NSS + 0.05 % gentamicin sulphate; or (2) the ovaries were washed three times with phosphate buffered saline (PBS) and transported in PBS + 0.05 % gentamicin sulphate. Samples of the ovarian cortex were obtained from each treatment group. Half of samples of each treatment was processed for histologic evaluation of follicular morphology and developmental stage using haematoxylin & eosin, and the other half was processed to analyse the ultrastructure of immature follicles using transmission electronic microscopy. The rates of good-quality follicles were 36.95 % and 88.6 % for OH + NSS and PBS, respectively. Histological analysis showed higher number of abnormal follicles in OH + NSS than in PBS treated ovaries (F1,12 = 99.64, P = 0.0001). The proportions of morphological alterations were not significantly different between treatments (χ21,7 = 7.72, P = 0.358). The ultrastructure of immature follicles from OH + NSS ovaries showed cell damage, revealed by abundant cell detritus around both, the oocyte and follicular cells. However, the presence of alterations was barely evident in both the cytoplasm and the nucleus of the granulosa cells.
Article Details
References
Van den Hurk R, Zhao J. Formation of mammalian oocytes and their growth, differentiation and maturation within ovarian follicles. Theriogenology. 2005;63:1717-51. doi: 10.1016/j.theriogenology.2004.08.005.
Fair T. Follicular oocyte growth and acquisition of developmental competence. Anim Reprod Sci. 2002;78:203-16. doi: 10.1016/S0378-4320(03)00091-5.
Asgari F, Valojerdi MR, Ebrahimi B, Fatehi B. Three dimensional in vitro culture of preantral follicles following slow-freezing and vitrification of mouse ovarian tissue. Cryobiology. 2015;71:529-36. doi: 10.1016/j.cryobiol.2015.11.001.
Silber S, Kagawa N, Kuwayama M, Gosden R. Duration of fertility after fresh and frozen ovary transplantation. Fertil Steril. 2010;94(6):2191-6. doi:10.1016/j. fertnstert.2009.12.073.
Valente B, Alvares P, Hayashi J, Lopes B, Colombo MH, Gaitkoski D, Basso AC, Arnold DR, Marcondes M. A new direct transfer protocol for cryopre-served IVF embryos. Theriogenology. 2016;85:1147-51. doi: 10.1016/j. theriogenology.2015.11.029.
Santos RR, Amorim C, Cecconi S, Fassbender M, Imhof M, Lornage J, Paris M, Schoenfeldt V, Martinez-Madrid B. Criopreservation of ovarian tissue: an emerging technology for female germline preservation of endangered species and breeds. Anim Reprod Sci. 2010;122:151-63. doi: 10.1016/j.anireprosci.2010.08.010.
Dittrich R, Lotz L, Keck G, Hoffmann I, Mueller A, Beckmann MW, Ven H, Mon-tag M. Live birth after ovarian tissue autotransplantation following overnight transportation before cryopreservation. Fertil Steril 2012;97(2):387-90. doi: 10.1016/j.fertnstert.2011.11.047.
Rodriguez C, Rocha V, Penitente JM, De Azevedo JL, Gomes R, Alves CA. The base medium affects ultrastructure and survival of bovine preantral fol-licles cultured in vitro. Theriogenology. 2016;85:1019-29. doi: 10.1016/j. theriogenology.2015.11.007.
Carvalho AA, Faustino LR, Silva CMG, Castro SV, Lopes CAP, Santos RR, Báo SN, Figueiredo JR, Rodrigues APR. Novel wide-capacity method for vitrifica-tion of caprine ovaries: Ovarian Tissue Cryosystem (OTC). Anim Reprod Sci. 2013;138:220-27. doi: 10.1016/j.anireprosci.2013.02.015.
Borges EN, Silva RC, Futino DO, Rocha-Junior CMC, Amorim CA, Báo SN, Lucci CM.Cryopreservation of swine ovarian tissue: Effects of different of cryopro-tectans on the structural preservation of preantral follicle oocytes. Cryobiology. 2009;59:195-200. doi: 10.1016/j.cryobiol.2009.07.003.
Albano E. Alcohol, oxidative stress and free radical damage. P Nutr Soc. 2006;65:278-90. doi: 10.1079/PNS2006496.
Hernández-Rodríguez S, Gutiérrez-Salinas J, García-Ortíz L, Mondragón-Terán P, Ramírez-García S, Núñez-Ramos NR. Estrés oxidativo y nitrosativo como me-canismo de daño al hepatocito producido por el metabolismo del etanol. Med Int Mex. 2014;(30):295-308.
Devine PJ, Perreault SD, Luderer U. Roles of reactive oxygen species and antioxidants in ovarian toxicity. Biol Reprod. 2012;86(2):27. doi: 10.1095/biolreprod.111.095224.
Amorim CA, Rodrigues APR, Rondina D, Gonçalves PBD, Figueiredo JR, Giorgetti A. Cryopreservation of ovine primordial follicles using dimethyl sulfoxide. Fertil Steril. 2003;79(1):682-86. doi: 10.1016/S0015-0282(02)04820-3.
Celestino J, Rodrigues R, Lopes C, Sousa F, Matos M, Melo M, Nair S, Ribeiro A, Viana J, Figueiredo J. Preservation of bovine preantral follicle viability and ultra-structure after cooling and freezing of ovarian tissue. Anim Reprod Sci. 2008;108:309-18. doi: 10.1016/j.anireprosci.2007.08.016.
Lopes CAP, Rodrigues APR, Celestino J, Melo M, Nogueira R, Cabral C, Viana J, Báo SN, Jewgenow K, Figueiredo J. Short-term preservation of canine prentral follicles: Effects of temperature, medium and time. Anim Reprod Sci. 2009;115:201-14. doi: 10.1016/j.anireprosci.2008.12.016.
Rodrigues APR, Amorim CA, Costa SHF, Matos MHT, Santos RR, Lucci CM, Báo SN, Ohashi OM, Figueiredo JR. Cryopreservation of caprine ovarian tissue using dimethylsulphoxide and propanediol. Anim Reprod Sci. 2004;84:211-27. doi: 10.1016/j.anireprosci.2003.12.003.
Cavalcante AYP, Gouveia BB, Barberino RS, Lins TLBG, Santos LP, Gonçalves RJS, Celestino JJH, Matos MHT. Short Communication: kit ligand promotes the transition from primordial to primary follicles after in vitro culture of ovine ovari-an tissue. Zygote. 2015;1-5. doi: 10.1017/S0967199415000556.
Santos RR, Tharasanit T, Figueiredo JR, Van Haeften T, Van den Hurk T. Pres-ervation of caprine preantral follicle viability after cryopreservation in sucrose and ethylene glycol. Cell Tissue Res. 2006;325:523-531. doi: 10.1007/s00441-006-0193-5.
Fransolet M, Labied S, Henly L, Masereel MC, Rozet E, Kirschvink N, Nisolle M, Munaut C. Strategies for using the sheep ovarian cortex as a model in re-productive medicine, PLoS One. 2014;9(3):e91073. doi: 10.1371/journal. pone.0091073.
Hayashi M, Hariya M, Kayano M, Suzuki H. Brief communication. Distribution of follicles in canine ovary - A simple and rapid method for counting follicles. Cryobiology. 2015;71:514-51. doi: 10.1016/j.cryobiol.2015.08.014.
Merchant-Larios H, Moreno-Mendoza N, Buehr M. The role of the mesonephros in cell differentiation and morphogenesis of the mouse fetal testis. Int J Dev Biol. 1993;37:407-15.
De Bruin JP, Dorland M, Spek ER, Posthuman G, Van Haaften M, Looman CWN, Te Velde ER. Ultrastructure of the resting ovarian follicle pool in healthy young women. Biol Reprod. 2002;66:1151-60. doi: 10.1095/biolreprod66.4.1151.
Paulini F, Carvalho R, Jivago JL, Lucci CM. Ultrastructural changes in oocytes during folliculogenesis in domestic mammals. J Ovarian Res. 2014;7:102. doi: 10.1186/s13048-014-0102-6.
Lucci CM, Rumpf R, Figuereido JR, Báo SN. Zebu (Bos indicus) ovarian pre-antral follicles: morphological characterization and development of an effi-cient isolation method. Theriogenology. 2002;57:1467-83. doi: 10.1016/S0093-691X(02)00641-6.
License

Veterinaria México OA by Facultad de Medicina Veterinaria y Zootecnia - Universidad Nacional Autónoma de México is licensed under a Creative Commons Attribution 4.0 International Licence.
Based on a work at http://www.revistas.unam.mx
- All articles in Veterinaria México OA re published under the Creative Commons Attribution 4.0 Unported (CC-BY 4.0). With this license, authors retain copyright but allow any user to share, copy, distribute, transmit, adapt and make commercial use of the work, without needing to provide additional permission as long as appropriate attribution is made to the original author or source.
- By using this license, all Veterinaria México OAarticles meet or exceed all funder and institutional requirements for being considered Open Access.
- Authors cannot use copyrighted material within their article unless that material has also been made available under a similarly liberal license.